Transfer of acquired products according to the FDA requirements
Development of over 60 technologies for pharmaceutical dosage forms
Process validation according to the EU and FDA requirements
GMP implementation in pharmaceutical production plants
Due diligence audit
GMP compliance audit
Suppliers audit
Site Master File preparation
Conceptual design for solid dosage forms, sterile and lyophilisation
plant, multi purpose drug factory, pharmaceutical warehouse, R&D
facility, QC laboratories etc.
Strategic development studies, site development study
Product development studies
Marketing analyses
Product registration
Management of three new drug factory construction
Numerous bidding procedures for pharmaceutical equipment clean rooms,
HVAC systems, laboratory equipment etc.
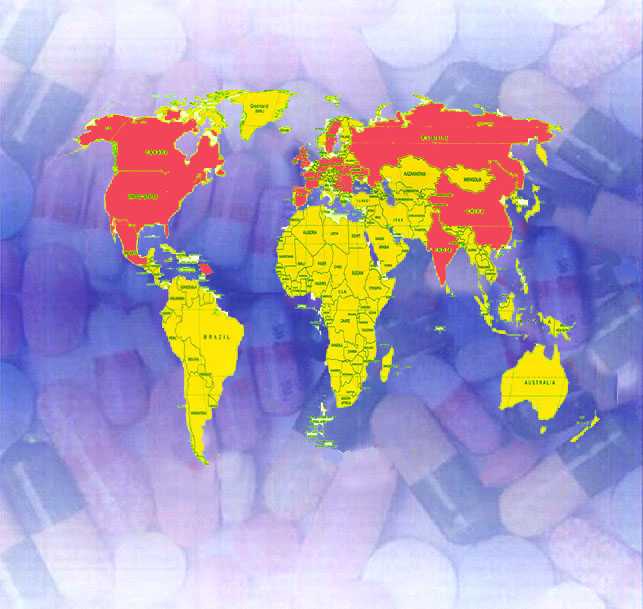
Working mainly for the big multinational company members of our consulting organization collected worldwide knowledge and experience in pharmaceutical industry. Countries in which members of our team have worked include:
America (USA, Puerto Rico, Canada, Mexico)
Western Europe (Spain, Switzerland, Scandinavia, UK, Ireland, Germany, France)
Central Europe (Poland, Hungary)
Eastern Europe (Russia, Yugoslavia, Macedonia, Ukraine, Bulgaria, Romania)
Asia (China, India, Dubai, Bangladesh)